Journal of
eISSN: 2572-8466


Research Article Volume 2 Issue 5
Department of Mechanical and Biomedical Engineering, City University of Hong Kong, Hong Kong
Correspondence: Kelvii Wei Guo, Department of Mechanical and Biomedical Engineering, City University of Hong Kong, 83 Tat Chee Avenue, Kowloon Tong, Kowloon, Hong Kong, Tel 852 3442 4621
Received: September 20, 2016 | Published: April 4, 2017
Citation: Guo KW. Property of zinc oxide (Zno) nanostructures potential for biomedical system and its common growth mechanism. J Appl Biotechnol Bioeng. 2017;2(5):197-202. DOI: 10.15406/jabb.2017.02.00046
Zinc Oxide (ZnO) nanomaterials have received broad attention thanks to their distinguished performance in electronics, optics and photonics. Due to the novel applications in optoelectronics, sensors, transducers and biomedical sciences, ZnO materials have become a leading edge in nanotechnology. Therefore, the properties of ZnO nanostructures potential for applying in biotechnology are marked along with the relevant common growth mechanism aim to provide the vital information about the growing field related to nano-ZnO in the biomedical system with environmental friendly nature and biocompatibility.
Keywords: nanostructures, nanoparticles, nanowires, nanorods, growth mechanism, biomedical system.
PL, photoluminescence; PLE, photoluminescence excitation; PVP, polyvinylpyrrolidone; MOCVD, metal organic chemical vapor deposition; VLS, vapor-liquid-solid; ZnO, zinc oxide
To date, zinc oxide (ZnO) has been a crucially versatile material in nanotechnology applications. It is well known that ZnO has a wide band gap (3.37eV) compound semiconductor attractively suitable for short wavelength optoelectronic applications1,2 and its high exciton binding energy (60meV) ensure efficient excitonic emission at room temperature, which already has been reported in published literature with disordered nanoparticles and thin films.3,4 Also, ZnO is transparent to visible light and can be made highly conductive by doping5-7 Moreover, due to its unique characteristics of crystal structure, the lack of a symmetry center in wurtzite structure and large electromechanical coupling result in strong piezoelectric and pyroelectric properties and show predominant applications in mechanical actuators and piezoelectric sensors8-10 chemical sensors11,12 and solar cells13,14
Wurtzite ZnO
Wurtzite ZnO has a hexagonal structure (space group C6mc) with lattice parameters a= 0.3296 and c= 0.52065 nm. The structure of ZnO can be simply described as a number of alternating planes composed of tetrahedrally coordinated O2- and Zn2+ ions, stacked alternately along c-axis as shown in Figure 115 The tetrahedral coordination in ZnO results in non-central symmetric structure and consequently piezoelectricity. More important characteristic of ZnO is its polar surfaces. The most common polar surface is the basal plane. The oppositely charged ions produce positively charged Zn-(0001) and negatively charged O-(000-1) surfaces resulted in a normal dipole moment and spontaneous polarization along the c-axis with a divergence in surface energy. To maintain a stable structure, the polar surfaces generally have facets or exhibit massive surface reconstructions, but ZnO± (0001) are exceptions: they are atomically flat, stable and without reconstruction.16 Efforts to understand the superior stability of the ZnO± (0001) polar surfaces are at the forefront of research in today’s surface physics.17,18 The other two most commonly observed facets for ZnO are {2-1-10} and {01-10}, which have non-polar surfaces and lower energy than the {0001} facets.
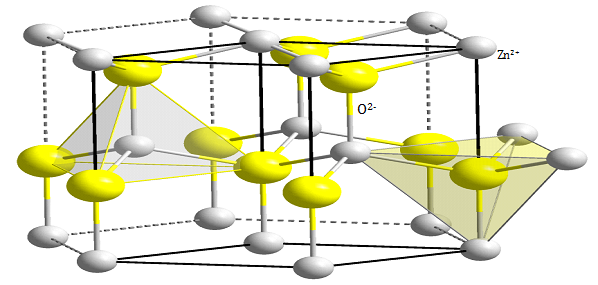
Figure 1 Wurtzite structure of ZnO .15
ZnO growth
For ZnO, there are three types of fast growth directions: <2-1-10> (±[2-1-10], ±[-12-10], ±[-1-120]); <01-10> (±[01-10], ±[10-10], ±[1-100]) and ±[0001]. ZnO exhibits a wide range of novel structures that can be grown by tuning the growth rates along these directions on the basis of its polar surfaces. A crystal has different kinetic parameters for different crystal planes, which are emphasized under controlled growth conditions. After nucleation and incubation, a crystallite will commonly grow into a 3D object with well-defined, low index crystallographic faces. Figure 2 shows a few typical ZnO growth morphologies of 1D nanostructure.19 These structures tend to maximize the areas of the {2-1-10} and {01-10} facets due to the lower energy. It shall be noted that planar defects and twins are observed occasionally parallel to the (0001) plane and dislocations are rarely seen.
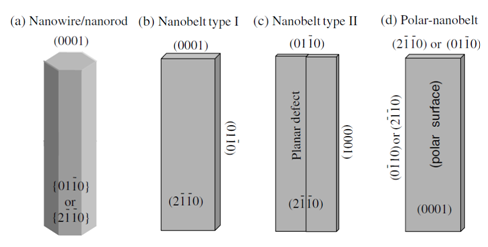
Figure 2 Typical growth morphologies of one-dimensional ZnO nanostructures and the corresponding facets.19
Property of ZnO nanostructures
Due to the quantum size effect, increasing band gap energy of 1D ZnO and nanoparticles can be proved by its photoluminescence property.20-22 X-ray absorption spectroscopy and scanning photoelectron microscopy reveal the enhancement of surface states with the downsizing of ZnO nanorods.23-25 Furthermore, the carrier concentration in 1D systems can be significantly affected by the surface states.26,27 Therefore, study of the properties of individual ZnO nanostructures is fundamentally essential to future innovative design of functional nanoscale devices.
Field emission property:The electric field emission from vertically aligned ZnO nanowires/nanorods has been extensively investigated for these years. One-dimensional (1D) nanomaterials with sharp tip are promising candidates for electron field emission thanks to their natural property from the geometry. Several groups have been reported the field emission properties from vertically aligned ZnO nanoneedles and nanowires.28-32 Results show that turn-on field was found to be ~18 V/mm at a current density of 0.01mA/cm2 and the emission current could reach 0.1mA/cm2 at 24 V/mm. The better results for ZnO nanowires synthesized at low temperature were obtained with the turn-on field of 6 V/mm at the current density of 0.1mA/cm2 and the emission current reached 1mA/cm2 at 11 V/mm, which could provide sufficient brightness as flat panel display. More compellingly, in the electron emission from ZnO nanotetrapod, a low turn-on field of 1.6 V/mm can be realized at a current density of 1mA/cm2. This improved emission performance was attributed to the higher aspect ratio of tetrapod structure than that of nanowires.33-36
Luminescence properties:ZnO exhibits a direct band gap of 3.37 eV at room temperature with a large exciton binding energy of 60 meV. Compare to other wide band gap materials (eg. GaN, 25meV),37,38 ZnO has much stronger exciton binding energy. The efficient exciton emission at room temperature can be easily triggered under low excitation energy because thermal energy of ZnO at room temperature is 26meV. As a consequence, ZnO is recognized as a promising photonic material in the blue-UV region. Single crystalline ZnO nanowires have been synthesized using high temperature VLS (vapor liquid solid) growth methods. Room temperature UV lasing in those well-aligned ZnO nanowires has been gained successfully. Results show that there are three emitting bands in PL (Photoluminescence) spectra of ZnO nanorods measured with the fluorescence at an excitation wavelength of 325 nm at room temperature, including a strong ultraviolet emission at around 386 nm and a very weak blue bane (440± 480 nm) as well as an almost negligible green band (510± 580 nm).39-41 For ZnO nanoparticles, the broad emission band located on the visible range is more obvious than that of the nanorod structure. There is also a PLE (Photoluminescence excitation) spectrum located on the UV region with a maximum intensity wavelength of 370 nm. It illustrates that the UV emission of ZnO is contributing to the near band edge emission while the green band emission corresponds to the surface defect. It demonstrates that oxygen vacancies are responsible for the green luminescence in ZnO. Oxygen vacancies occur in three different charge states: the neutral oxygen vacancy (V0O), the singly ionized oxygen vacancy (V*O) and the doubly ionized oxygen vacancy (V**O), for which only V*O can act as the so-called luminescent centers.42-45
ZnO nanostructures
Up to now, one-dimensional (1D) ZnO nanostructures have been considered to be one of the most important nanomaterials for fabricating nanodevices with applications in optics, electronics, mechanics and biomedical sciences, especially in the system of bioimaging/biosensor and drug/gene delivery.
ZnO nanoparticles:ZnO nanoparticles have received attractive and intensive attention for recent years because of its unique optical properties. Nano ZnO crystals show a compelling and convincing luminescence behavior in the visible region. Meanwhile, the emission wavelengths can be tuned by changing the nanoparticles size. More attractively, ZnO is a promising candidate for the environmental friendly nature and biocompatibility in the bio-medical system, especially for bio-imaging.40,46,47 Also, the exceptional optical and electrical properties of ZnO nanoparticles may arise from the confinement of excitons and phonons in nanostructures in three dimensions. Although it is reported that the Bohr exciton radius of ZnO is about 2.34 nm, the size-dependent optical property (band gap variation) of ZnO nanoparticles, for the ones which are much larger than the quantum confinement size, are still be investigated to the large scale.48,49 In the past decades, various methods have been employed to produce photoluminescence ZnO nanoparticles.50-53 Depending on the fabrication techniques and the surface quality of ZnO nanoparticles, the origin of UV photoluminescence in ZnO nanoparticles is either recombination of confined excitons or surface-bound ionized acceptor-exciton complexes. Furthermore, more and more unique behaviors are continuously being explored. However, colloidal ZnO nanocrystals obtained from sol-gel route tend to aggregate due to their surface energy. As a result, such nanocrystals are unstable in aqueous dispersion during storage. To stabilize ZnO nanoparticles with surface modifications effective in stabling luminescent ZnO colloids and often well dispersed in organic solvents, various capping agents, such as amines, long chain aliphatic thiols and Polyvinylpyrrolidone (PVP), have been employed.54-58 Nevertheless, the majority of bio-analyses require water-dispersible materials and in this regard, synthesis of water-stable ZnO nanoparticles with emission in visible region is still needed to investigate further.
ZnO nanowires and nanorod:Since the successful growth of aligned ZnO nanowires on a single-crystal substrate, a system essentially useful for vertical device fabrication has been found. As a result, great interest in acquiring more control over the alignments, including supporting substrates, distribution of nanowires and density of nanowires, to maximally meet the requirements of nanodevices has been inspired.59,60
Mechanism of common ZnO nanostructures growth
Aligned growth of ZnO nanowires can be achieved by various methods, such as vapor-liquid-solid (VLS) process.61,62,63 metal-organic chemical vapor deposition (MOCVD),64-67 sol-gel process68-70 and aqueous chemical growth process.63,71 As follows, two commonly used mechanisms are expressed.
Vapor Liquid Solid (VLS) growth mechanism:Vapor liquid solid (VLS) growth process is a commonly used method to synthesize one-dimensional nanostructures.72 This mechanism was first proposed by Wagner and Ellis in 1964 for the growth of Si whisker.73 Results show that Si whickers with diameters up to micrometer were grown by hydrogen reduction of SiCl4. Au, Pt, Ag, Pd, Cu and Ni played the roles as the catalysts in this growing process. In the VLS process, metal catalyst and the desired material vapor form an alloy and liquefy at the reaction temperature. The metal catalyst can be sensibly chosen from the phase diagram by identifying metals in which the nanowires component elements are soluble in the liquid phase but do not form solid compounds more stable than the desired nanowires phase. Au, Cu, Co, Fe and Ni are those commonly used catalysts. The function of the catalyst is to form a crystallization site of nanowires. During the nanowires growth process, catalyst and nanowires material form a low melting point eutectic alloy where is a preferential site for absorption of gas-phase reactant, supersaturated and nucleation for crystallization. Nanowire growth follows by the supersaturating and nucleation of liquid alloy and continues as long as the catalyst alloy remains in a liquid state and the reactant is available. The catalyst droplet also determines the diameter and direction of the growing nanowires. Ultimately, the growth terminates when the temperature is below to the eutectic temperature of the catalyst alloy, or the reactant is no longer available. The VLS growth mechanism of ZnO nanorods is shown schematically in Figure 3.74 Based on the relationship among major parts of growth system, a conceptual model has been summarized in Figure 4.
Aqueous chemical growth mechanism:Aqueous chemical growth technique has been developed to produce functionalized thin films and coatings of metal oxide materials on various substrates such as amorphous, single crystalline, polycrystalline and flexible substrates. This method is appealing because it is catalyst-free, low cost, low growing temperatures and good potential for scale-up. In addition, this synthetic strategy also does not require any certain template, surfactant membrane or epitaxial substrate to control their orientation.75,76 Recently, various nanostructures are synthesized successfully on the basis of aqueous chemical growth method, such as nanorods,77,78 nanowires,79,80 nanoflowers,81,82 nanoneedles.83 nanoplates,84 nanosheets ,85 nanotowers 24 and nanotubes.86 The synthesis involves the hydrolysis-condensation of hydrated metal ions and complexes (inorganic polymerization) and their hetero nucleation onto substrates. Experimentally, the aqueous chemical growth simply consists of heating an aqueous solution with the metal precursors (salts or complexes) at a given pH value and ionic strength, in the presence of mild temperatures (below 100°C). This synthetic process requires neither expensive and complicated setups nor hazardous high-pressure containers. Moreover, the organic-free and low temperature approach makes this process safe and environmental friendly. Thanks to the absence of organic solvent, the purity of the as-prepared thin films is substantially improved, that is, the residual salts can be easily removed by water. In most cases, no additional heat or chemical treatments are the superior approach compared to the sol-gel process as well as surfactant, template and membrane-based synthesis methods. The utilization of temperature and chemical sensitive substrates required by certain applications is therefore allowed. The processes were focused on controlling the reaction time, concentration of solution, different pH values and reagents to produce ZnO nanostructures with different morphologies, especially for the capping reagents, which are remarkable to control the shapes of ZnO nanostructures successfully.
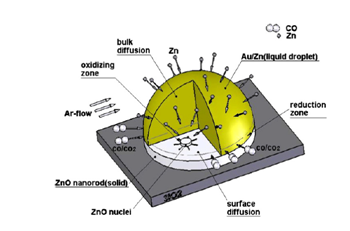
Figure 3 Schematic illustration of nucleation and growth of ZnO nanorods by the VLS mechanism; Zn atoms condense and attach to the edges of nuclei and then oxidized by CO/CO2; lateral growth of ZnO nuclei causes the completion of one ZnO monolayer.74
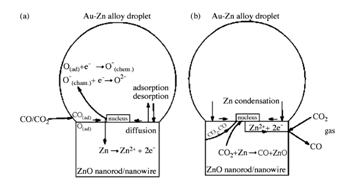
Figure 4 Conceptual model of the VLS growth of the ZnO nanorods
The current status of the use of ZnO nanomaterials for biomedical applications in biomedical imaging, drug delivery, gene delivery and biosensor is flourishing. Consequently, for the environmental friendly nature, greener and simpler methods for ZnO growth will be investigated along with the detailed mechanism. Meanwhile, with the tremendous achievements of ZnO nanomaterials in biomedical systems coupled with its attractive properties, much research effort will be needed to develop biocompatible/biodegradable ZnO nanomaterials for the ultimate responsibilities.
None.
The author declares no conflict of interest

©2017 Guo. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.