International Journal of
eISSN: 2573-2889


Mini Review Volume 8 Issue 1
1Department of Health Sciences, Federal University of Espírito Santo, Brazil
2Department of Structural and Functional Biology, Institute of Biology, University of Campinas, Brazil
3Department of Education and Humanities, Federal University of Espírito Santo, Brazil
Correspondence: Pedro Víctor Carvalho, Department of Health Sciences, Federal University of Espírito Santo, UFES Campus São Mateus Highway BR101 Km 60, São Mateus, Espírito Santo, Brazil, Tel +552733121582
Received: August 04, 2025 | Published: August 29, 2025
Citation: Carvalho PV, Carvalho MGS, Silva MM. GLP-1 agonists in pediatric obesity: an integrative perspective on cell signaling, development, and metabolism. Int J Mol Biol Open Access. 2025;8(1):53-56. DOI: 10.15406/ijmboa.2025.08.00198
Pediatric obesity is an escalating public health concern with long-lasting metabolic, inflammatory, and psychosocial consequences (Graphical abstract). Glucagon-like peptide-1 receptor (GLP-1R) agonists, such as liraglutide and semaglutide, have emerged as therapeutic options in pediatric populations due to their effectiveness in promoting weight loss and improving glycemic control. This mini-review examines the cellular and molecular effects of these agents in children and adolescents, with a focus on key intracellular signaling pathways activated by GLP-1R stimulation, and the potential impacts on cell differentiation, tissue remodeling, bone development, and the gut microbiota. A critical analysis of recent literature highlights the importance of integrative and cautious approaches that account for the developmental plasticity of the pediatric organism and the possible pleiotropic effects of early pharmacological intervention.
Keywords: GLP-1, pediatric obesity, cell signaling, development, microbiota, molecular pharmacology
GLP-1, glucagon-like peptide-1; GLP-1R, glucagon-like peptide-1 receptor; BMI, body mass index; CNS, central nervous system; BDNF, brain-derived neurotrophic factor; AKAP, A-kinase anchoring protein; cAMP, cyclic adenosine monophosphate; PKA, protein kinase A; CREB, cAMP response element-binding protein; PI3K, phosphoinositide 3-kinase; AKT, protein kinase B; MAPK, mitogen-activated protein kinase; ERK, extracellular signal-regulated kinase; Epac, exchange protein directly activated by cAMP; Wnt, wingless/integrated signaling pathway; β-catenin, beta-catenin; SCFAs, short-chain fatty acids.
The prevalence of obesity among children and adolescents has increased globally in recent decades. Beyond behavioral and environmental factors, biological components such as neuroendocrine dysfunction, chronic inflammation, and altered metabolism actively contribute to the pathophysiology of excess weight in this age group. In this context, glucagon-like peptide-1 receptor (GLP-1R) agonists have been increasingly used in the treatment of pediatric obesity, with evidence supporting their efficacy in reducing body weight, BMI, and improving glycemic control, although gastrointestinal side effects, such as nausea, are common.1,2 These drugs act on both the central nervous system and the gastrointestinal tract, promoting satiety and reducing food intake, as demonstrated by brain imaging studies using GLP-1-specific tracers.3 Additionally, they appear to modulate systemic inflammation via the gut–adipose tissue–brain axis, with increased levels of beneficial microbes such as Akkermansia muciniphila and production of metabolites like inosine, which improve leptin sensitivity and reduce inflammation.4 A relevant interaction is also observed between GLP-1R agonists and the gut microbiota, although the specific effects of GLP-1 receptor signaling on this axis remain only partially elucidated. Studies in animal models suggest that diet-induced obesity alters intestinal microbial composition and reduces endogenous GLP-1 levels, reinforcing the interdependence of these systems.5 In terms of safety, population-based data indicate a favorable neuropsychiatric profile for GLP-1R agonists, with a lower risk of suicidal ideation among adolescents using these drugs compared to controls.6 Nevertheless, while clinical outcomes are promising, the underlying molecular mechanisms—including receptor signaling, neuroendocrine regulation, and long-term developmental effects—require further investigation to support robust hypotheses.7
The GLP-1 receptor (GLP-1R) is a G protein-coupled receptor that, when activated, triggers complex intracellular signaling cascades. Key pathways include cAMP/PKA/CREB, PI3K/AKT, and MAPK/ERK. As illustrated in Figure 1, activation of the cAMP/PKA/CREB axis promotes the differentiation of white adipocytes into thermogenic-like adipocytes and enhances brown adipose tissue activity, contributing to lipid metabolism regulation and energy balance8. In hippocampal neurons, this same pathway induces BDNF expression, suggesting potential neurotrophic and metabolic roles during development.9 In pancreatic β-cells, GLP-1R signaling recruits contact proteins between the endoplasmic reticulum and mitochondria, such as AKAPs, thereby supporting mitochondrial remodeling and cellular energy homeostasis, processes that are particularly relevant during development (Figure 1, center).10 In mesenchymal stem cell models, the GLP-1R–PKA–β-catenin axis has been shown to modulate the balance between adipogenesis and osteogenesis, indicating a direct impact on cell differentiation and tissue remodeling.11 In cardiac tissues, GLP-1R activation reduces oxidative stress and mitochondrial dysfunction via the cAMP/Epac/PI3K/AKT pathway, with implications for cell survival and stress adaptation.12 In pancreatic β-cells, signaling mediated by β-arrestin 2 activates the MAPK/ERK pathway and CREB at pharmacological levels, influencing insulin secretion and metabolic adaptation (Figure 1, right).13 Given these multiple actions, in pediatric models, GLP-1R activation may directly interfere with metabolic plasticity, cellular differentiation, and tissue remodeling, requiring caution regarding potential off-target effects during growth and development.
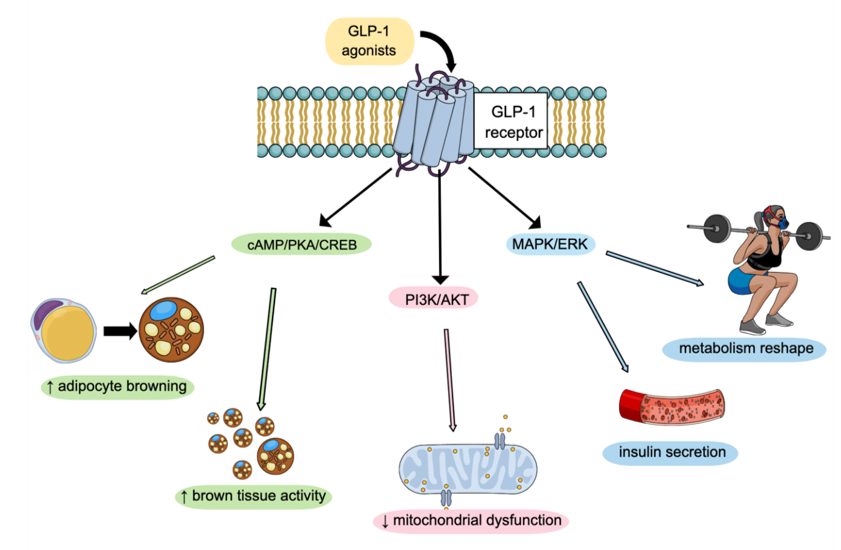
Figure 1 Schematic representation of GLP-1 receptor (GLP-1R) signaling pathways and their metabolic effects.
Upon binding of GLP-1 receptor agonists, GLP-1R activates multiple intracellular cascades, including cAMP/PKA/CREB, MAPK/ERK, and PI3K/AKT. These pathways promote adipocyte browning, increase brown adipose tissue activity, improve mitochondrial function, and regulate insulin secretion. In parallel, systemic effects involve metabolic reshaping and enhanced energy expenditure, positioning GLP-1R signaling as a central regulator of metabolic adaptation in obesity.
The gut microbiota is increasingly recognized as a central regulator of energy balance and metabolic health in children. In pediatric obesity, dysbiosis is frequently observed and is associated with altered nutrient absorption, increased energy harvest, and heightened inflammatory signaling, thereby reinforcing weight gain and metabolic dysfunction.14–16 Beyond compositional shifts, the functional consequences of dysbiosis are particularly relevant: the microbiota contributes to obesity pathophysiology by driving the production of short-chain fatty acids (SCFAs), modulating bile acid metabolism, and influencing immune and inflammatory pathways.15–17 Early-life determinants, which includes mode of delivery, infant feeding practices, and antibiotic exposure, could shape microbial communities and are strongly linked to later obesity risk, underscoring the long-term impact of microbiota development on metabolic outcomes.16,18 Importantly, recent findings reveal a bidirectional interaction between the gut microbiota and the GLP-1/GLP1R axis, positioning this interface as a key mediator of metabolic regulation in childhood (Figure 2). On the one hand, microbial metabolites such as short-chain fatty acids and secondary bile acids stimulate GLP-1 secretion from intestinal L-cells and modulate its circadian rhythm, linking microbial activity to satiety regulation, glucose control, and fat metabolism (Figure 2, left).19–21 On the other hand, pharmacological activation of GLP-1R reshapes the gut microbial community.22–24 GLP-1R agonists have been shown to enrich beneficial taxa such as Akkermansia muciniphila and Bifidobacterium spp., changes that correlate with improved glycemic responses and reduced systemic inflammation (Figure 2, left).25,26 Together, these findings highlight the gut microbiota-GLP-1 axis as a central node in pediatric obesity pathophysiology and therapy. Microbiota composition not only contributes to disease risk but may also predict responsiveness to GLP-1R agonists, suggesting that future therapeutic strategies could combine microbiota-targeted approaches with GLP-1R modulation to optimize outcomes in children and adolescents.
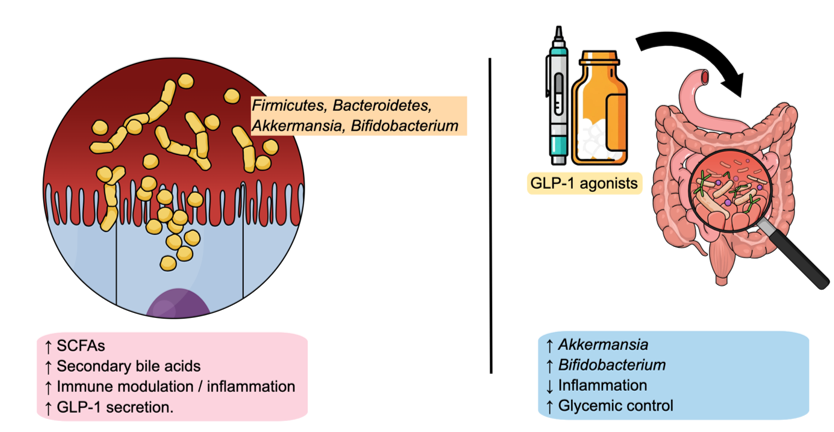
Figure 2 Bidirectional interactions between gut microbiota and GLP-1 receptor agonists in metabolic regulation. Gut microbiota composition (including Firmicutes, Bacteroidetes, Akkermansia, and Bifidobacterium) influences host metabolism by producing short-chain fatty acids (SCFAs), generating secondary bile acids, modulating immune and inflammatory pathways, and stimulating GLP-1 secretion from intestinal L-cells. Conversely, treatment with GLP-1 receptor agonists reshapes the microbial community, enriching beneficial taxa such as Akkermansia and Bifidobacterium, while reducing inflammation and improving glycemic control.
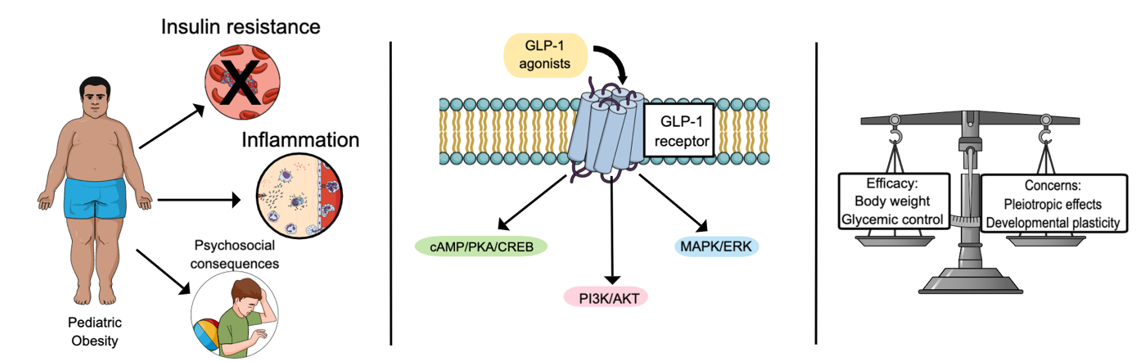
Graphical abstract Pediatric obesity is associated with insulin resistance, chronic inflammation, and psychosocial consequences. GLP-1 receptors (e.g., liraglutide and semaglutide) act through GLP-1 receptors activating intracellular signaling pathways (cAMP/PKA/CREB, PI3K/AKT, and MAPK/ERK) which regulate metabolic, proliferative, and differentiation processes. While these agents show great therapeutic efficacy in improving body weight and glycemic control, their use in developing organisms raises concerns about pleiotropic effects and impairment in developmental plasticity, requiring integrative approaches and cautious evaluation. Figure was created by the author.
In the therapeutic landscape of pediatric obesity, GLP-1R agonists have emerged as particularly effective agents when compared with other available strategies. Drugs such as semaglutide, liraglutide, exenatide, and dulaglutide have been shown to significantly reduce body weight, BMI, and BMI z-score in children and adolescents, with semaglutide demonstrating the greatest efficacy among the class.27 In addition, GLP-1R agonists improve glycemic control, especially in individuals with insulin resistance or type 2 diabetes, further reinforcing their clinical utility.28 By contrast, lifestyle interventions, including dietary modification, increased physical activity, and behavioral therapy, remain the cornerstone of first-line treatment.27 These approaches are most effective when sustained and family-based, offering long-term benefits for overall health. However, many children fail to achieve sufficient or sustained weight loss through lifestyle changes alone, highlighting the need for adjunctive therapies. Other pharmacological options, such as orlistat, phentermine, and the phentermine-topiramate combination, are approved for pediatric use but generally yield less weight reduction than GLP-1R agonists. These agents are also associated with distinct side effect profiles, which may limit tolerability and adherence. Bariatric surgery, reserved for the most severe cases, is highly effective in achieving substantial and durable weight loss. Nonetheless, its invasive nature, perioperative risks, and the potential for long-term nutritional deficiencies raise important concerns, particularly when performed during critical developmental stages.29 In this context, GLP-1R agonists represent an intermediate therapeutic option, more effective than lifestyle or other pharmacological approaches, yet less invasive than surgical procedures. This positioning highlights their growing relevance as a bridge between conservative and invasive treatments, offering a balance between efficacy, safety, and feasibility in pediatric obesity management.
During childhood and adolescence, several molecular pathways involved in energy regulation also participate in cellular growth and maturation. Inflammatory metabolic pathways such as PI3K-Akt have been longitudinally associated with bone mineral density in adolescents, indicating that their pharmacological modulation may interfere with critical periods of bone formation.30 Furthermore, metabolic conditions such as iron deficiency can alter progenitor cell fate, favoring adipogenesis over osteogenesis, underscoring the sensitivity of these developmental processes to energetic signals.31 Obesity and metabolic stress also impair bone development during adolescence, with evidence that excess adiposity and poor glycemic control compromise bone mass acquisition, potentially through inflammatory and epigenetic mechanisms.32 More intense metabolic interventions, such as bariatric surgery in adolescents, result in significant and sustained reductions in bone mineral density, highlighting the structural costs of chronic manipulations of energy regulatory systems during vulnerable stages.33,34 Bone-derived signals, such as osteocalcin (a non-collagenous protein secreted by osteoblasts) and sclerostin (a glycoprotein produced by osteocytes), maintain a bidirectional dialogue with energy metabolism and may influence systemic homeostasis, producing trans-systemic effects during growth.35 The Wnt/β-catenin pathway, essential for bone formation, also affects adipose tissue fibrosis and energy metabolism, illustrating how bone-targeted interventions may lead to unexpected systemic consequences.36 Although no major qualitative sex differences have been identified in the mechanisms of action of GLP-1 receptor agonists, quantitative variations in treatment response and adverse effects have been reported, potentially influenced by interactions with sex hormones such as estrogens.29,37 The biological basis of these differences remains poorly understood, and current evidence is largely derived from adult populations. In pediatrics, most available studies do not stratify outcomes by sex, underscoring an important knowledge gap and the urgent need for research that addresses sex-specific responses during childhood and adolescence. In this context, pharmacological modulation of energy homeostasis pathways may exert pleiotropic effects, including body mass reduction accompanied by decreased bone density, interference with neuronal and adipogenic precursor differentiation, and epigenetic modifications with potential transgenerational impact. These risks may be further modulated by sex-specific biological factors, which remain poorly characterized in pediatric populations. Together, these aspects warrant caution in the chronic use of GLP-1 receptor agonists during periods of active growth and maturation, reinforcing the need for long-term, sex-stratified studies to ensure safe and effective therapeutic application.
Despite the demonstrated efficacy of GLP-1 receptor agonists in reducing weight and improving metabolic control in adolescents, their long-term molecular and systemic consequences remain poorly understood. Given the complex interplay between metabolism, bone development, cell differentiation, and the gut microbiota during growth, these pharmacological interventions must be applied with caution and supported by robust longitudinal evidence. Moreover, quantitative sex-related differences in therapeutic response and adverse effects highlight the need for sex-stratified analyses, which are still lacking in pediatric studies. When compared to other strategies, GLP-1R agonists occupy an intermediate position, more effective than lifestyle or other pharmacological interventions, but less invasive than bariatric surgery, underscoring their unique therapeutic niche. Ultimately, pediatric obesity management should extend beyond weight reduction to address developmental safety, microbiota-endocrine interactions, and sex-specific variability, with molecular and integrative approaches paving the way for safer, more effective, and ethically sustainable protocols.
None.
The authors declared that there are no conflicts of interest.

©2025 Carvalho, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.