International Journal of
eISSN: 2381-1803


Research Article Volume 11 Issue 6
1Department of Biological and Biomedical Sciences, the Aga Khan University, Pakistan
2Bahauddin Zakariya University, Pakistan
3Department of Pharmacy, Capital University of Science and Technology, Pakistan
4King Saud bin Abdulaziz University for Health Sciences, Saudi Arabia
Correspondence: Samra Bashir, Professor, Department of Pharmacy, Capital University of Science & Technology, Islamabad Expressway, Kahuta Road, Zone-V, Islamabad, Pakistan, Tel +9251111555666
Received: October 11, 2018 | Published: November 20, 2018
Citation: Bashir S, Abbas S, Khan A. Pharmacological studies on prokinetic and laxative effects of trianthema portulacastrum linn. Int J Complement Alt Med. 2018;11(6):368-373. DOI: 10.15406/ijcam.2018.11.00428
Background: The current investigation on Trianthema portulacastrum Linn. (T. portulacastrum) was undertaken to provide the pharmacological basis for its medicinal use as a laxative.
Method: T. portulacastrum whole plant extract (Tp. Cr) was studied for in vivo laxative activity on mice and effect on isolated tissues preparations to find the mechanistic basis.
Results and discussion: Tp. Cr dose-dependently (10 and 30mg/kg) increased both total and wet fecal output in mice, similar to castor oil, indicating the presence of the laxative effect. In spontaneously contracting isolated rabbit jejunum, Tp. Cr (1-10mg/ml) caused pyrilamine sensitive spasmogenic effect, which was reproducible in guinea-pig ileum. On activity-guided fractionation, only aqueous fraction exhibited the stimulant activity, whereas the organic fractions caused an inhibitory effect on spontaneous and K+ (80mM)-induced contractions of rabbit jejunum, without causing any stimulation.
Conclusion: This investigation, indicating that T. portulacastrum whole plant possesses laxative activity possibly mediated through histaminergic mechanism, provides the scientific basis for the medicinal use of T. portulacastrum in constipation.
Keywords: Trianthema portulacastrum, laxative, spasmogenic, isolated gut preparations
Trianthema portulacastrum Linn. (Family; Aizoceae) is generally known as horse purslane and locally in Pakistan as itsit and biskhapra. The plant grows throughout Pakistan in crop fields, gardens, river side’s and waste lands as a fast-growing wild weed during the rainy season.1,2 It is a small glabrous prostrate herb and its parts have various medicinal uses. In traditional systems of medicine, the whole plant of T. portulacastrum is used as analgesic, laxative, stomachic, anti-asthmatic, for the treatment of bronchitis, itch, inflammation, piles, liver diseases, chronic ulcers, night-blindness and blood and skin diseases.3 T. portulacastrum root has been shown to possess cathartic, abortifacient and irritant properties and effective against cough, phlegmatic fevers and asthma. Leaves, due to their diuretic property, are recommended in oedema and dropsy while leaf extract is used as antidote to alcohol intoxication, to treat rheumatism and as anthelmintic medicin.4‒6
Experimental studies on various extracts and phytoconstituents of T. portulacastrum have confirmed the presence of a number of medicinal activities in this plant. T. portulacastrum has been reported to possess antilithic,7 antihyperglycemic,8 antibacterial and antifungal,9,10 hepatoprotective,11 anthelmintic,12 antihyperlipidemic,13,14 and central analgesic15 activities. The protective effect of T. portulacastrum on experimental hepatocarcinogenesis has also been established.16 Qualitative analysis for various classes of constituents has detected the presence of steroids, flavonoid, alkaloids, carbohydrates, tannins and terpenes in T. portulacastrum. Ecdysterone has been identified as the principal constitituent of T. portulacastrum.2 The other phytoconstituents isolated from T. portulacastrum include trianthenol,9 5,2'-dihydroxy-7-methoxy-6, 8-dimethylflavone and leptorumol.17 The plant also contains potassium salts, punarnavine, ecdysterone, and trianthemine.16 T. portulacastrum root contains saponin glycosides.18 Vanillic acid, ferrulic acid, P-hydroxybenzoic acid, trans-cinnamic acid, protocatechuic acid, o-coumaric acid, caffeic acid and pyrogallic acid are reported to be present in stem and leaves of the plant.19 T. portulacastrum has been used in indigenous systems of medicine to relieve constipation, however, the scientific data rationalizing its traditional use and defining the precise mode of action for such effects is scarce. This study was therefore designed to provide pharmacological basis for the medicinal use of T. portulacastrum as laxative.
Plant material, extraction and fractionation
The whole plant of T. portulacastrum, collected from the fields of Multan, was identified by an expert taxonomist of the Institute of Pure & Applied Biology, Bahauddin Zakariya University, Multan. The plant specimen has been deposited at herbarium of the same institute vide voucher no P. Fl 235-3. The plant material was washed to remove dirt and then completely dried in a shaded area. Subsequently, 200g of the dried plant was coarsely ground using electrical grinder and the material, taken into amber colored glass bottles, was soaked in aqueous-methanol (70% v/v) with occasional shaking for three days. The material was then filtered through a double layered muslin cloth to remove vegetative debris. The obtained filtrate was re-filtered through a filter paper.20 Extraction of the plant residue was repeated twice to ensure maximal extraction of the soluble constituents. Solvent from the combine filtrates was removed by evaporation at 37ºC under reduced pressure (-760mmHg) on a rotary evaporator (R-210, BUCHI, Switzerland). The thick, semi-solid paste of dark brown colour left after evaporation of the solvent represented the crude extract of T. portulacastrum whole plant (Tp. Cr), yielding 11%. Tp. Cr was made soluble in normal saline for the in vivo and 10 % DMSO for the in vitro experiments.
To fractionate the crude extract, aqueous solution of Tp. Cr (10 g in 150ml of water) was taken into a separating funnel. An equal volume of ethyl acetate was then incorporated to the separating funnel and the immiscible liquids were shaken together (3-5min). Separating funnel was then allowed to stand till the immiscible liquids separated into layers. The upper layer, constituting of ethyl acetate, was collected in a flask, while the aqueous layer was re-introduced into the separating funnel along with another volume of fresh ethyl acetate and the process was repeated for a total of three times. Evaporation of the solvent on a rotary evaporator from combined ethyl acetate layers so collected yielded the ethyl acetate fraction (Tp. EtAc), weighing 1.18g. The aqueous layer left after ethyl acetate treatment was similarly extracted with n-butanol to obtain n-butanol fraction (Tp. But), yielding 0.87g. The aqueous solution left subsequent to the extraction with n-butanol was similarly dried to obtain the aqueous fraction (Tp. Aq), weighing 7.2g. Normal saline solution was used to prepare stock solution of Tp.Aq, whereas, 10% aqueous dimethyl sulfoxide solution was used to prepare Tp. EtAc and Tp But stock solutions. The stocks of the crude extract and the fractions were freshly diluted in normal saline to the required concentrations on the day of experiment.
Drugs and chemicals used
Acetylcholine chloride (Ach), atropine sulphate, histamine diphosphate and pyrilamine maleate were purchased from Sigma Chemical Co., St Louis, Mo, USA. Reagent grade chemicals including potassium chloride, ethylenediamine tetra-acetic acid (EDTA), calcium chloride, glucose, magnesium chloride, magnesium sulphate, potassium dihydrogen phosphate, hydrochloric acid, sodium dihydrogen phosphate, sodium bicarbonate, sodium hydroxide, sodium chloride and ammonium hydroxide were used for making physiological salt solutions and were purchased from BDH Laboratory Supplies, Poole, England. Physiological salt solutions were prepared fresh in distilled water on the day of experiment. The drugs were solubilized in normal saline solution and the vehicle had no effect on tissue contractility in the control experiments. Both stock solutions of the drugs and their dilutions were freshly prepared on the day of experiment.
Animals
Rabbits, guinea-pigs and mice of local breed and either sex, used in this study, were housed in the Animal House of the Aga Khan University, Karachi under standard housing conditions (23-25ºC temperature and 12h light/dark cycle). The animals were provided tap water and standard diet. The animals were fasted for 24h prior to the experiments but had access to water throughout. To isolate the tissues, rabbits and guinea-pigs were stunned by a blow on back of the head and then sacrificed by cervical dislocation. Laxative effect of the plant material was studied on mice. Rulings of the Institute of Laboratory Animals Resources, Commission on Life Sciences, National Research Council21 were followed for ethical considerations and the experimental protocols were approved by Institutional Animal Ethics Committee, Bahauddin Zakariya University, Multan.
Laxative activity
According to a previously used method,22 mice were individually kept in the blotting sheet lined cages to count the fecal matter with convenience. Group 1, the negative control group, was treated with normal saline (10ml/kg), group 2, the positive control group, was administered with castor oil (10ml/kg), while groups 3, 4 and 5, the test groups, were given 10, 30 and 100mg/kg of Tp. Cr, respectively. Control substances as well as Tp. Cr were administered orally to the mice with the help of feeding needle. Data on fecal production (number of total as well as wet feces) from all the five groups were recorded till 18h post treatment.
Experiments on gut preparations
Experiments on isolated gut were performed according to the previously employed methods in our laboratory.23,24
Guinea-pig ileum
The ileum was dissected from guinea-pig and was kept in Tyrode’s solution. To study the effect of the drugs, 2cm long ileac segments were individually suspended in Tyrode’s solution filled tissue baths of 10ml capacity. Tyrode’s solution was aerated with carbogen (95% O2 and 5% CO2) and maintained at 37ºC. The composition of the Tyrode’s solution in mM was: KCl 2.68, NaCl 136.9, MgCl2 1.05, NaHCO3 11.90, NaH2PO4 0.42, CaCl2 1.8 and glucose 5.55. The suspended tissue segments were given initial tension of 0.7g and left untreated for at least 30min to get equilibrated with the environment. A guinea-pig ileum preparation, under such experimental conditions, does not exhibit any intrinsic contractions and is therefore considered suitable for studying spasmogenic activity.25 Once equilibrated, each tissue segment was repeatedly treated with Ach at sub-maximal concentration of 0.3µM every 3min until amplitude of the successively recorded contractile responses of the tissues became constant. Various bolus concentrations of the extract were then applied on resting tension of the tissue to determine the stimulant effect and the responses were measured as percent of the control drug-induced maximum stimulant effect.
Rabbit jejunum
Jejunum was dissected from rabbit and kept in Tyrode’s solution to clean off mesenteries. Jejunum was segmented in 2cm long pieces which were then suspended in tissue baths filled with carbogen aerated Tyrode’s solution at 37°C. Intrinsic intestinal activity and the drug responses were recorded using isotonica Transducer (Model 50-6360, Harvard Apparatus, Holliston, USA) coupled with bridge amplifier and PowerLab 4/25 data acquisition system attached to computer running Chart 6 software (AD Instruments, Sydney, Australia). A tension of 1g was then applied to each tissue segment and the tissues were left untreated for at least 30min to get equilibrated with the environment. Once equilibrated, each tissue segment was stabilized by treating with Ach at 0.3μM concentration every 5-10min till amplitude of the stimulant responses to Ach became identical. To test any modulatory effects of the plant material on jejunum motility, increasing concentrations of the plant extract were applied in a cumulative manner. Stimulant effect of the plant material was measured as percent of Ach-induced maximum contraction.
Statistical analysis
In this study, each experiment was repeated 3-4times and the data is expressed as mean±standard error of mean (S.E.M.) or the median effective concentration (EC50) with 95% confidence intervals (CI). Concentration–response curves (CRCs) were analyzed by non-linear regression using Graph Pad program (Graph PAD, San Diego, CA, USA). The statistical parameter applied is Student’s t-test but for laxative activity one-way ANOVA with Dunnett’s test as post hoc was used for multiple comparisons of the laxative effect of different doses of Tp. Cr with control. P<0.05 was considered statistically different.
Laxative effect
Tp. Cr administration to naïve mice orally, increased the total and wet feces produced over 18h after the administration (Table 1). The laxative effect was evident at both 10 and 30mg/kg doses where the lower dose increased the wet fecal count only, while the higher dose increased total fecal output without causing any further change in the consistency of feces. Tp. Cr at 100mg/kg dose did not affect the feces production as compared to the saline treatment. The standard laxative agent, castor oil, also increased both the wet and the total fecal output.
Effect on guinea pig ileum
Tp. Cr caused a concentration-dependent spasmogenic effect on guinea pig ileum at 0.01 to 10.0mg/ml, when applied in bolus concentrations (Figure 1A) (Figure 1B). Magnitude of the spasmogenic effect was 14.29±1.40, 14.88±3.82, 31.71±0.73, 84.04±5.50, 103.38±2.18, 103.70±1.09, 109.87±1.02, and 73.95±1.71 % (n=2), as compared to Ach (10µM) and 5.94±2.65, 11.28±4.18, 27.07±6.85, 72.06±6.75, 98.61±2.26, 101.4±3.30, 104.28±2.91 and 82.51±3.62 % (n=5), as compared to histamine (10µM)-induced maximum contractile effect, at 0.01, 0.03, 0.1, 0.3, 1.0, 3.0, 5.0 and 10.0mg/ml, respectively. Atropine (0.1µM) pretreatment had no significant inhibitory effect on the contractile responses (Figure 1A), while pyrilamine (1.0µM) pretreatment abolished the responses almost completely (Figure 1B).
Effect on rabbit jejunum
Tp. Cr induced a concentration-dependent (0.1-10.0mg/ml) spasmogenic response in the spontaneously contracting rabbit jejunum preparations (Figure 2A), followed by the relaxant effect in tissue segments which were left unwashed for some time. Both spasmogenic and spasmolytic effects were reproducible. Magnitude of the spasmogenic effect at 0.1, 0.3, 1.0, 3.0, 5.0 and 10.0mg/ml respectively was 2.54±1.48, 3.96±0.97, 17.66±7.53, 35.14±7.56, 71.36±14.63 and 120.43±14.13 % (mean±S.E.M., n=4) of Ach (30µM)-induced highest contraction.
Atropine pretreatment (0.1µM) did not block Tp. Cr-induced contractile responses of the tissue (data not shown); while in tissues pretreated with pyrilamine (1.0µM), the contractile effect was significantly suppressed (Figure 2A). Tp. Cr also inhibited high K+ (80mM)-induced contraction in rabbit jejunum preparations with EC50 value of 2.18 mg/ml (95% CI, 1.74-2.74, n=4). Verapamil in a concentration-dependent fashion inhibited high K+-induced contraction with median inhibitory concentration of 0.05µM (0.04-0.06, n=5), as depicted by Figure 2B. Tp. Cr also shifted Ca2+ CRCs towards right in a concentration-dependent (0.1-1.0mg/ml) manner (Figure 3A), similar to that caused by verapamil (Figure 3B).
Effect of the fractions on rabbit jejunum
Both Tp. EtAc and Tp But relaxed spontaneous as well as high K+-induced contractions without showing any stimulant activity in rabbit jejunum Figure 4A & 4B. However, Tp But was more potent against high K+ than the spontaneous contraction, a trend similar to that exhibited by verapamil (Figure 4C), whereas, Tp. EtAc relaxed the two contractions in an opposite fashion. Tp. Aq exhibited stimulant effect on spontaneously contracting rabbit jejunum which was quantified as 4.3±1.71, 36.35±5.97, 67.5±5.31, 101±13.21, 114±11.79% of Ach (30µM)-induced maximum contraction, at 0.3, 1, 3, 5 and 10mg/ml concentrations (Figure 5).
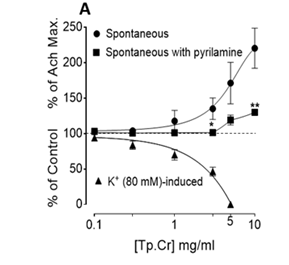
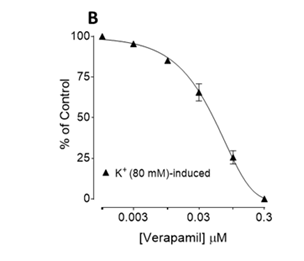
Figure 2 Effect of crude extract of Trianthema portulacastrum (Tp.Cr) on spontaneous contractions, in the absence and presence of pyrilamine, and K+ (80 mM)-induced contraction of isolated rabbit jejunum and (B) concentration-dependent inhibitory effect of verapamil on K+ (80 mM)-induced contraction of isolated rabbit jejunum preparations. Values are shown as mean±S.E.M, n=3-4 (* P<0.05, **P<0.01, Student’s t-test).
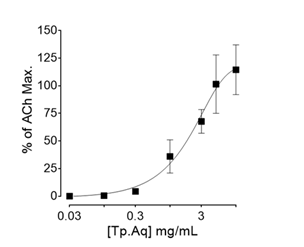
Figure 5 Stimulant effect of Trianthema portulacastrum aqueous fraction (Tp. Aq) on spontaneously contracting rabbit jejunum. Values are expressed as mean±S.E.M, n=2.
Group no. |
Treatment |
Dose |
Defecation/group (Mean±SEM) |
wet feces/group (Mean±SEM) |
% of wet feces/group (Mean±SEM) |
1 |
Saline |
10 |
9.5±2.06 |
0 |
0 |
2 |
Castor oil |
10 |
15.2±1.51* |
12.2±1.56** |
83.60±4.5** |
3 |
Tp.Cr |
10 |
9.6±1.02 |
3.8±1.11* |
27.34±5.7** |
4 |
30 |
15.4±1.43* |
4.2±0.84* |
38.23±4.25** |
Table 1 Laxative activity of the crude extract of Trianthema portulacastrum (Tp.Cr)
*P<0.05, **p<0.01 vs. saline treated groups N=5
Increase in the global interest in traditional medicine has led to the accelerated efforts to scientifically monitor and regulate traditional medicine including herbal drugs.26 Due to folkloric reputation of T. portulacastrum Linn. In hypomotility disorders of gut, the possible laxative effect of the crude extract was studied on mice. When administered at 10 and 30mg/kg doses to the healthy animals, the plant extract produced a dose-dependent laxative effect, which was manifested as increase in fecal output with a mild decrease in fecal consistency. In comparison, castor oil, a known laxative agent, caused increase in fecal output which largely constituted of wet feces. Castor oil causes effect on gastrointestinal motility and secretions through its hydrolytic product, ricinoleic acid,27 which changes electrolytes and water transportation through epithelial lining of gut resulting in increased bulk of the intestinal content and accelerated peristaltic movements of the colon. Results of this study suggest that T. portulacastrum extract possesses laxative effect comparable to castor oil which is mediated mainly through increase in gastrointestinal motility than increase in secretions.
Coordinated contraction and relaxation of longitudinal and circular smooth muscles of gastrointestinal wall, responsible for the gut motility, is regulated by neural and hormonal factors. An alteration in the normal gastrointestinal motility may cause various major disorders such as diarrhea, spasm, and constipation. Increased gut motility, through rapid intestinal transit of the content, may result in diarrhea. Spasm is sudden and unorganized contraction of muscles in the gastrointestinal wall, which is often associated with pain and interference of the function. Constipation results from hypo motility of gut and occurs because of various muscular and neurologic disorders.27
Isolated tissue preparations are widely used to explore mechanistic basis of a give pharmacological effect.22,28 Application of the crude extract on spontaneously contracting isolated rabbit jejunum caused concentration-dependent spasmogenic activity, which was however transient and eventually superseded by the relaxant effect in the tissues if left unwashed for some time. The contractile effect of Tp. Cr was partially blocked in tissues pre-incubated with pyrillamine (1.0µM) but remained unaltered in the presence of atropine (0.1µM). This suggests the presence of constituents holding histaminergic activity in the plant extract and accounting for the spasmogenic effect at least in part. The observed spasmogenic effect was further tested on guinea-pig ileum to assess reproducibility of the effect with specie variation and to quantify the contractile responses because, being a quiescent gut preparations, guinea-pig ileum is perceived useful for this purpose.25 The maximum spasmogenic response of the crude extract was comparable to histamine-induced maximal contraction. Pretreatment of the tissues with pyrilamine, (1.0µM), a histamine receptor (H1) antagonist,29 abolished the extract-induced contractile effect, while atropine, a muscarinic antagonist,30 did not alter it, which shows that Tp. Cr causes gut stimulation through histaminergic pathway. Literature evidence strongly suggests the role of histamine as an important cellular messenger in gastrointestinal tract,31 which causes contractions of the smooth muscles by acting on H1-receptors.32
To evaluate the possible nature of gut inhibitory constituents, the plant extract was tested on rabbit jejunum pre-contracted with high K+. High K+ induces contractile responses in smooth muscles by increasing intracellular Ca2+ through internal release and extracellular Ca2+ influx through L-type voltage-sensitive channels.33 Therefore, a substance causing inhibition of high K+ -induced contractions is regarded a blocker of calcium influx.34 The contractile effect elicited by high K+ was completely inhibited by Tp. Cr, suggesting the calcium channel blocking (CCB) effect in T. portulacastrum. The finding was confirmed when pre-incubation of the tissue with Tp. Cr shifted Ca2+ CRCs towards right in a concentration-dependent manner, similar to the standard calcium-channel blocker, verapamil.35 Calcium antagonists, including verapamil, constitute an important therapeutic group. These drugs are characterized by their concentration-dependent inhibitory effect on slow entry of Ca2+ and reversal of the effects caused by Ca2+.36
This natural existence of gut inhibitory and stimulatory effects combination in T. portulacastrum is probably meant to limit spasmogenic effect to therapeutic requirement, extending to which it may cause abdominal cramps, as seen with chemical drugs used in constipation.27 These investigations indicate that T. portulacastrum whole plant possesses laxative effect mediated through histaminergic mechanism, which provides a scientific basis for its folkloric use in constipation.
This study was supported in part by research fund given to Dr. Samra Bashir by Department of Biological and Biomedical Sciences, the Aga Khan University, Karachi, Pakistan.
The authors declare that there is no conflict of interest.

©2018 Bashir, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.