eISSN: 2576-4462


Research Article Volume 9 Issue 1
1Agricultural Engineer, Plant Sciences Department, Ponta Grossa State University, Brazil
2PhD., Plant Sciences Department, Ponta Grossa State University, Brazil
3Professor, PhD., Plant Sciences Department, Ponta Grossa State University, Brazil
Correspondence: Ricardo Antonio Ayub, Professor, PhD., Plant Sciences Department, Ponta Grossa State University, Brazil
Received: December 20, 2024 | Published: January 16, 2025
Citation: Mello G, Reis CA, Ayub RA. Different IBA concentrations in the production and quality of Buxus sempervirens seedlings. Horticult Int J. 2025;9(1):7-10. DOI: 10.15406/hij.2025.09.00316
The Boxwood plant (Buxus sempervirens), a member of the Buxaceae family, is widely valued for its use in landscaping and topiary. This study investigates the effect of different concentrations of indole-3-butyric acid (IBA) on Buxus sempervirens cuttings, propagated vegetatively. Conducted at the Viveiro Florestal of the State University of Ponta Grossa, the experiment tested survival rates, rooting, new shoot production, and biomass characteristics. No significant differences were found for most parameters, except for the dry mass of aerial and total parts, where 2 g/L of IBA yielded the highest averages.
Keywords: IBA, auxin, boxwood propagation, adventitious roots, cutting method
The species Buxus sempervirens, popularly known as boxwood, belongs to the Buxaceae family and originates from North Africa, Europe and Asia. The Buxus genus has the largest number of species within its family, between 95 and 100 species.1,2 Mainly used as landscape plants, hedges, ornamentals or as topiary.3-5 For its propagation, the vegetative propagation method can be cited, which is the propagation of a plant from a tissue, cell or organ, in the case of roots, stems, branches or leaves.6 This propagation occurs because each plant cell is totipotent, which has the genetic information necessary to generate a new plant.7-9
The rooting of cuttings is affected by internal and external conditions, in which internal conditions are related to the morphology and physiology of the plant, and external factors to environmental conditions.10 An excellent resource to enhance rooting success is the use of phytoregulators, which are plant hormones that regulate plant development and growth.11,12 They can originate naturally, originating in leaf primordia, in the case of indoleacetic acid (IAA), or synthesized, in the case of auxins such as Idol-3-Butyric Acid (IBA), which bring greater results in the formation of adventitious roots of both stem and leaf cuttings due to their ability to stimulate plant elongation.13-15 The objective of this work was to evaluate the influence of different concentrations of auxin (IBA) on the rooting process combined with the quality of seedling production, environmental conditions of the region and plant age, in response to the propagation and survival of the Buxus sempervirens species.
The experiment was conducted on the premises of the Forest Nursery of the State University of Ponta Grossa (UEPG), between February 2022 and June 2022 in the municipality of Ponta Grossa (Paraná, Brazil), under the coordinates of Latitude 25º05'44” S, Longitude 50º06'29” W and altitude of 912 meters. According to the Köeppen classification, the climate of the region is of the Cfb type, characterized as humid subtropical, with mild summers and an average temperature of around 18°C, with a maximum of 24°C and a minimum of 13°C. The average annual precipitation for the region is around 1,500 mm to 1,800 mm, with rainfall evenly distributed throughout the year, with frost occurring in autumn and winter.16 (Figure 1)
Plants
The mother plants of the Buxus semprervirens species were established in field conditions, under commercial management and with quarterly fertilization of 50 grams of NPK (10-10-10) per plant. The shoots were collected in the morning, placed in polystyrene boxes, moistened and transported to the experimental site. Cuttings averaging 6 cm in length were prepared from the collected shoots, cut in a bevel at the base and straight above the last apical bud, maintaining all the original leaves.
Chemical products
Then, the bases of the cuttings were treated with the synthetic phytoregulator Indole-3-Butyric Acid (IBA) at treatment concentrations of 0, 1, 2 and 3 g/L in a hydroalcoholic solution (50% v/v), with 10 seconds of immersion.
The cuttings were then planted in 200-cell trays, with an individual volume of 0.0125 cm3, filled with Carolina Soil Padrão® substrate, with a composition of 85% Sphagnum Peat and 25% Vermiculite, with the addition of 2 kg.m-3 of controlled-release fertilizer (FLC) from the Osmocote® brand (18-05-09; 5 months).
After 60 days, the seedlings began to receive the application of a nutrient solution composed of 1.2 g/L of MAP, 4 g/L of Urea, 4 g/L of Potassium Chloride, 1 g/L Dipsol® Micro (3.4% Fe, 0.85% B, 1.1% Mg, 0.5% Cu, 0.05% Mo, 3.2% Mn, 4.2% Zn), applied every 15 days with a solution volume of approximately 5 liters for 1000 seedlings.
Seedling quality
The cuttings were planted at a depth of approximately 1 cm and then stored in a greenhouse with humidity above 90% and without temperature control. After 60 days in the greenhouse, the seedlings were transferred to the nursery greenhouse for 30 days, where they received micro-sprinkler irrigation with nebulizers, with an individual flow rate of 33 liters per hour. After the acclimatization period, the seedlings were transferred to the shade house for 30 days, where they received micro-sprinkler irrigation with an individual flow rate of 165 liters per hour, totaling 120 days.
Evaluations performed
The evaluations on the Buxus sempervirens propagules were performed 120 days after planting, which were evaluated in percentage of survival propagules, mortality, roots with length up to the bottom of the cell, new shoots, number of shoots per cutting and moisture content. The dry mass of the aerial part, roots and total were also evaluated.
To obtain the evaluation of the moisture content and the dry mass of the aerial part, roots and total, the following were performed: After 120 days, a sample of five cuttings was obtained by repeating all treatments, randomly chosen using the random function in the Microsoft Excel® application. The roots were washed in running water to remove the substrate (Figure 2) and dried naturally to eliminate excess water. The samples were then weighed to determine their green mass, on a scale with a precision of 0.01 g. Immediately after weighing, they were placed in properly identified paper bags and left in a drying oven at 60ºC for 72 hours. They were then weighed again as total dry mass. The moisture content found in the samples was calculated as a percentage using the difference in percentage of green mass (100%). The dry mass of the aerial part, roots and total in grams was also obtained by weighing.
Statistics
The experiment was set up according to a completely randomized design, with four treatments corresponding to the IBA concentrations, with the concentrations of each treatment being respectively 0 g/L, 1 g/L, 2 g/L and 3 g/L, with four replicates per treatment and 100 propagules per sample unit. The variances of the treatments were assessed for homogeneity by the Bartlett test (p<0.05) and the variables that presented significant differences (p<0.05) in the analysis of variance (ANOVA) had their means compared by the Tukey test at 5% probability. The regression analysis of the means was then assessed if they were significant (p<0.05) and the best-fitting trend line was used. The statistical analyses were performed using the R Studio software.
The variables in the results can all be considered normal and homogeneous according to the Shapiro-Wilk test and the Barlett's test at 5% significance. In relation to the evaluations of survival percentage, propagules with roots up to the bottom of the cells, new shoots and the average number of new shoots per Buxus sempervirens propagule (Table 1), there were no significant differences according to the statistical tests in the analysis of variance (ANOVA) or the regression test. The dependent variables did not respond to variations in the independent variable. In the regression test, the inflection point of the respective variables was 2.78 g/L for survival, 1.45 g/L for mortality and 2.78 g/L for roots with length to the bottom of the cells (Table 1).
|
Present |
Concentration of AIB |
|||
|
|
0g/L |
1g/L |
2g/L |
3g/L |
|
Survival |
80,75 ns |
68,5 ns |
76,75 ns |
82,75 ns |
|
Mortality |
19,25 ns |
31,5 ns |
23,25 ns |
17,25 ns |
|
Roots with length up to the botton of the cells |
80,75 ns |
68,5 ns |
76,75 ns |
82,75 ns |
|
New shoots |
32 ns |
28,75 ns |
29,75 ns |
26,5 ns |
|
Average number of new shoots per propagule |
2,75 ns |
2,5 ns |
2,5 ns |
2,2 ns |
Table 1 Percentage of survival, mortality, roots with length to the bottom of the cells, new shoots and the average number of new shoots per propagule of Buxus sempervirens in different concentrations of AIB in the city of Ponta Grossa - PR
*Survival values show CV = 17.59%, mortality shows CV = 59.53%, roots at the bottom of the cells CV = 17.59%, new shoots CV = 31.74% and average number of new shoots per plant CV = 21.6%. According to the Shapiro-Wilk test and the Bartlett test at 5% significance, the means of the variances can be considered normal and homogeneous. According to the F test, the means cannot be considered different. ns = not significant
The survival of cuttings may be related to their initial reserve and not to the addition of phyto-regulators, varying according to the cutting technique and the degree of lignification of each species or cutting.17
These responses show that, regardless of the concentration of IBA used, Buxus sempervirens cuttings have a high capacity to form new adventitious roots, proving that rooting does not differ due to the supply of exogenous auxins. Another reason why there was no difference in rooting between the treatments may be due to the fact that only young propagules were used for vegetative propagation, because according to Stuepp18 studies show that young propagules have higher rooting percentages compared to propagules with advanced ontogeny (Figure 3) or because the propagules have sufficient amounts of endogenous auxins for rooting.19
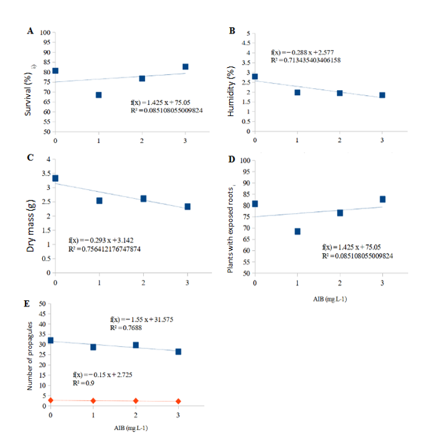
Figure 3 Regression analysis: A) Percentage of plant survival; B) Moisture content in the cuttings; C) Dry mass; D) Plants with apparent roots; E) Number of Buxus semperviren propagules in different concentrations of IBA in the city of PontaGrossa –PR after 120 days of planting.
In this study, we can see that the percentage of roots differed more when compared to the percentage of new shoots, indicating that in the Buxus sempervirens species, the formation of the root system has a greater allocation of reserves than the formation of the aerial part. According to Delgado and Yuyama20 in their study with woody cuttings of Myrciaria dubia, they showed that the species has high rooting rates due to the fact that root formation occurs before the shoots, which allows the cuttings to allocate their reserves to root formation rather than new shoots.
In relation to the analysis of the moisture content and dry mass of the root part of the propagules (Table 2), there were no significant differences in the statistical tests in the analysis of variance (ANOVA), and also by the regression test (Figure 2). As for the dry mass of the aerial and total parts (Table 2), there were significant differences in the statistical tests in the analysis of variance (ANOVA), with treatment 3 (2 g/L) being significant in both cases, but in relation to the regression test there were no significant differences (Figure 3). In relation to the regression test, the inflection point for the respective variables was 2.4 g/L for the moisture content, 2.34 g/L for the dry mass of the aerial part, 2.28 g/L for the dry mass of the root part and 2.08 g/L for the total dry mass.
|
Present |
Concentration of AIB |
|||
|
|
0g/L |
1g/L |
2g/L |
3g/L |
|
Moisture content |
63,76 ns |
61,34 ns |
64,49 ns |
66,08 ns |
|
Dry mass of aerial part |
1,85 b |
1,95 b |
2,8 b |
1,98 b |
|
Dry mass of root part |
0,53 ns |
0,55 ns |
0,68 ns |
0,48 ns |
|
Total dry mass |
2,38 b |
2,5 ab |
3,48 a |
2,45 b |
Table 2 Percentage of moisture content, weight in grams of the dry mass of aerial, root and total cuttings of Buxus semperviren in different concentrations of IBA in the city of Ponta Grossa- PR after 120 days of planting.
*Values for moisture content show CV = 9.97%, aerial part dry mass CV = 17.49%, root part dry mass CV = 23.92% and total dry mass CV = 17.4 %. Means followed by the same letter in the row do not differ by the T-test at 5% probability. According to the F test, the averages for moisture content and root dry mass were not different. ns = not significant.
The moisture content values in the plants indicate how important it is to use nebulizers to control the irrigation of the cuttings and maintain an adequate level. According to Oliveira21 in his technical recommendation on rooting cuttings for seedling production, he explains the importance of using misting systems to reduce water loss from the cuttings, favoring the hydration of the substrate and controlling the temperature. He recommends placing the tubes or cells in greenhouses with intermittent misting or micro-sprinkler irrigation so that the internal humidity remains above 80%.
In this study, even though there was a significant difference in the average dry mass of the aerial part and the total, we can hypothesize that this influence is not due to the action of the different concentrations of IBA, because the highest average was not in the highest concentration, but because, according to Bortolini22 and Alcântara,23 cuttings accumulate reserves, mainly carbohydrates, in the fall and winter, altering the mass of the aerial part.
It is important to note that cuttings may have sufficient levels of endogenous auxin in their tissues to promote the formation of new roots.11 According to the same author, the lack of response to the action of exogenous auxin in some species may be due to the high lignification of the cuttings, hindering its action. He therefore states that the exogenous application of growth regulators to vegetative propagules is not significant in some cases. Botin and Carvalho24 state that auxins are growth regulators that induce the formation of adventitious roots in cuttings, but in some cases they have little or no effect on the species.
The rooting of cuttings is affected by internal and external conditions. In the present study, Buxus sempervirens seedlings were propagated in controlled environments using a synthetic phytoregulator, specifically Idol-3-Butyric Acid (IBA), which has the role of stimulating greater results in the formation of adventitious roots of both stem and leaf cuttings. In the present study, no responses were obtained in the use of IBA auxin for propagation by cuttings of Buxus sempervirens. In the quality of seedling production with the control of planting, humidity and fertilization, similar effects were obtained in relation to survival, rooting, new shoots, moisture content and total dry mass of the cuttings when compared without the use of the synthetic phytoregulator.
None.
All authors declare that there is no conflicts of interest.

©2025 Mello, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.