eISSN: 2378-315X


Research Article Volume 12 Issue 4
PCOM School of Pharmacy/Wellstar North Fulton Hospital/ NGOC Roswell, USA
Correspondence: Sonia Amin Thomas, PCOM School of Pharmacy/Wellstar North Fulton Hospital/NGOC Roswell, Georgia, USA
Received: July 06, 2023 | Published: July 16, 2023
Citation: Thomas SA, Ling H. Evaluation of granulocyte-colony stimulating factor use in the outpatient infusion center setting. Biom Biostat Int J. 2023;12(4):95-98. DOI: 10.15406/bbij.2023.12.00390
Background: Neutropenic fever remains a serious complication of chemotherapy due to the myelosuppressive effects of most antineoplastic regimens. Granulocyte colony-stimulating factors (G-CSFs) have been effective in reducing the risk of developing febrile neutropenia and decreasing its duration. However, most patients with a low risk of FN who use GCSFs may incur unnecessary costs, possible adverse effects, and may have fewer benefits than those seen in intermediate and high-risk patients.
Objective: The purpose of this study is to determine if G-CSF medications are being utilized in accordance with the National Comprehensive Cancer Network (NCCN) guidelines for primary or secondary prevention of febrile and non-febrile neutropenia due to chemotherapy within the network of physicians groups and infusion centers.
Study design and methods: This is a multi-center, retrospective, chart review over a 3-month period. The data was collected from patients seen at any of the Northwest Georgia Oncology Centers (NGOC), Wellstar Medical Group, and LaGrange infusion center locations from June 1, 2020 to August 31, 2020. Patients aged 18 and older with a cancer diagnosis who received either pegfilgrastim, filgrastim, or a biosimilar were included in the analysis. The patient charts were reviewed for the presence of chemotherapy regimens and the subsequent G-CSF use in addition to any patient specific risk factors for febrile neutropenia.
Results: A total of 283 patients met the inclusion criteria and were evaluated. The average age was 61 years old, and the patient population was 75.3% female. Of the patients evaluated, 268 out of 283 patients (95%) utilized either pegfilgrastim, filgrastim, or a biosimilar in accordance with the NCCN guidelines for prevention or treatment of neutropenia or febrile neutropenia due to chemotherapy.
Conclusion: We found that over the three-month study period, the majority of G-CSF medication administrations were used in accordance with the NCCN guidelines in the setting of oncology patients within the healthcare management group.
Key words: GCSF, neutropenic fever, pegfilgrastim, filgrastim, chemotherapy
NCCN, national comprehensive cancer network; G-CSF, granulocyte colony-stimulating factors; FN, febrile neutropenia; NGOC, north georgia oncology clinics; eGFR, estimated glomerular filtration rate; ANC, absolute neutrophil count
Neutropenic fever remains a serious complication of oncologic chemotherapy due to the myelosuppressive effects of many antineoplastic regimens.1 The mortality rate of neutropenic fever is between 5% and 13% for most patients, but approaches higher percentages for patients with multiple risk factors.1 In addition, it is associated with longer hospitalizations, serious infections, and increased medical costs.2 Granulocyte colony-stimulating factors (G-CSF) have been effective in reducing the risk of developing febrile neutropenia and decreasing its duration.2
The G-CSF medications used in the North Georgia Oncology Clinic (NGOC), Wellstar Medical Group, and the LaGrange Infusion Center are filgrastim and pegfilgrastim, both available as subcutaneous injections. Filgrastim requires multiple days of therapy, whereas pegfilgrastim is available is a one-time subcutaneous injection.3 A new formulation of pegfilgrastim includes an auto-injector in which the dose is administered automatically approximately 27 hours after it is applied to the patient.4
The NCCN categorizes most chemotherapy regimens into various risk categories for developing febrile neutropenia, denoted high-, intermediate-, or low-risk. The current NCCN guidelines recommend G-CSF use for primary and secondary prevention of neutropenic fever. G-CSF medications are recommended in patients receiving high-risk chemotherapy, defined as a 20% or greater risk of developing febrile neutropenia (FN), as a category 1 recommendation. G-CSF medications are also recommended for intermediate-risk patients with comorbidities that place them at higher risk for febrile neutropenia. This is further defined as a risk of developing febrile neutropenia between 10-20% with at least one of the following conditions: prior chemotherapy or radiation, persistent neutropenia, bone marrow involvement, recent surgery or open wounds, liver dysfunction, renal dysfunction, or age greater than 65 years receiving full-dose chemotherapy. Patients receiving chemotherapy with less than 10% risk of developing febrile neutropenia are considered low-risk, and G-CSF therapy is not recommended in these patients for primary prevention, though they may be used for secondary prevention.3
In clinical practice, the NCCN guidelines have not always been followed, leading to prescribing G-CSF for patients with a low risk of developing febrile neutropenia.1 In a study completed by Waters and colleagues in 2013, overutilization of pegfilgrastim was reported to be present in up to 46% of chemotherapy patients with risk of FN less than 20%.5 A study completed by Hanna et al also showed that 47% of filgrastim and 8.7% of pegfilgrastim uses did not meet the criteria of >20% risk of febrile neutropenia nor did they meet the criteria for intermediate risk with comorbidities.2 Inappropriate use of filgrastim and pegfilgrastim may contribute to potentially unnecessary adverse effects and cost to the patient.2
There is less data looking into stewardship of supportive care medications in chemotherapy, and this area may have opportunity for cost-savings. The purpose of this study is to evaluate the use of filgrastim and pegfilgrastim in the outpatient cancer center to determine if the use of G-CSF medications is being utilized in a way that is in accordance with the NCCN guidelines for patients utilizing them for primary or secondary prevention of febrile neutropenia.
The primary objective of this study is to evaluate the use of G-CSF medications for primary or secondary prevention of neutropenic fever at multiple infusion center locations which included the Wellstar North Georgia Oncology Clinics (NGOC), Wellstar Medical Group, and the LaGrange Infusion Center. The secondary objectives were to determine if use of G-CSFs was appropriate based on chemotherapy risk level as defined by the NCCN, appropriate when selected for clinic location, and to determine the estimated cost of inappropriate G-CSF use.
This was a multi-center, retrospective, chart review over a 3-month period. The study was approved by the Wellstar IRB committee. Patients were selected via a medication use report of pegfilgrastim or filgrastim over the specified time period. The sample size was 320 patients. Patients were included if they were seen at any Wellstar North Georgia Oncology Clinic (NGOC), Wellstar Medical Group (WMG), and the LaGrange Infusion Center from June 1, 2020 to August 31, 2020, were 18 years or older, had a cancer diagnosis, and received pegfilgrastim, filgrastim, or a biosimilar agent for either primary or secondary prevention of febrile neutropenia. Patients were excluded if they did not have a cancer diagnosis, or if a colony stimulating factor was used for an indication other than prevention or treatment of neutropenic fever in chemotherapy patients.
The data collected included age, weight, gender, cancer diagnosis, chemotherapy regimens, estimated glomerular filtration rate (eGFR), total bilirubin, absolute neutrophil count (ANC), white blood cell count, and the colony stimulating factor used. The chart was also reviewed for any history of neutropenia or neutropenic fever as documented by an absolute neutrophil count below 1000 cells/mm3 or if a previous episode was documented the chart. If there was no documentation of a neutropenic episode or history of neutropenia, or any other risk factor, the use was deemed inappropriate. A descriptive analysis including frequencies and percentages of all requested variables was performed, using mean and standard deviation, as appropriate.
The total number of patient records obtained in this study was 320. Of the 320 patient charts examined, 37 patients were excluded for the absence of a cancer diagnosis or administration record of a G-CSF during the time period. The total number of patients that received at least one dose of a G-CSF during the study period was 283 (Table 1). Among them, there were 213 female patients and 70 male patients. Patient demographics were 194 white, 61 African- American, 11 Hispanic/Latino, 6 Asian, and 11 other or nondisclosed, and the average age of the patient population was 60.3 +/- 2.8 years (Table 1). The most common cancer in this study was breast cancer (Table 1). The number of patients receiving high-risk chemotherapy was 148 (52.3%), and the number of patients receiving intermediate-risk or low-risk chemotherapy was 123 patients (43.5%) and 12 patients (4.2%), respectively. The majority of patients (98.6%) received pegfilgrastim or a pegfilgrastim biosimilar as the G-CSF.
G-CSF medications were used appropriately in all patients receiving high-risk chemotherapy, 113 out of 123 patients (91.9%) receiving intermediate-risk chemotherapy, and in 7 out of 12 patients (58.3%) receiving low-risk chemotherapy (Figure 1). The types of cancers with inappropriate use were non-small cell lung cancers, diffuse large B-cell lymphoma, uterine cancer, and small cell cancer. The chemotherapy regimens with inappropriate G-CSF use were all classified as the intermediate- or low-risk for febrile neutropenia.
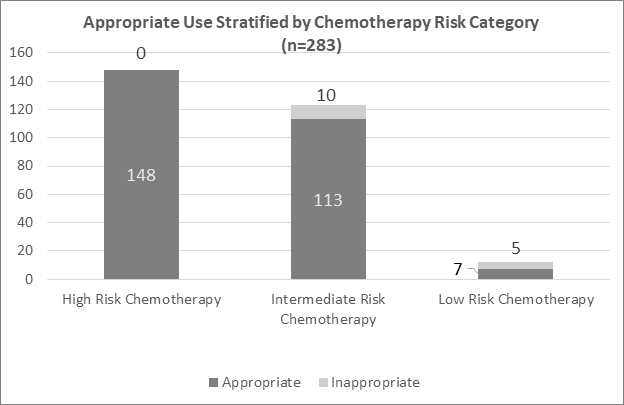
Figure 1 Appropriate Use of G-CSF Stratified by risk category of chemotherapy.
When stratified by risk category, high-risk chemotherapy had no inappropriate uses, intermediate-risk consisted of 10 of 123 patients (8.13%) with inappropriate use, and low-risk chemotherapy consisted of 5 out of 12 patients with inappropriate use (41.6%).
The most common reason for inappropriate use was an absence of an indication in the notes or no history of febrile or dose-limiting neutropenia, occurring in 11 out of 15 patients that received a G-CSF outside of guideline recommendations. There were two patients who received a G-CSF for prevention of COVID-19 infection while receiving chemotherapy. This was deemed inappropriate because it is not recommended by NCCN and may be associated with a higher risk of worsening clinical condition in the case of COVID-19.6 Other inappropriate uses included a cancer that did not involve bone metastases, and one patient had discontinued doxorubicin in the RCHOP regimen, which reduced their neutropenia risk from high to intermediate with no other risk factors present.3 The criteria for appropriate use in patients receiving intermediate-risk chemotherapy regimens was made to be intentionally broad to account for physician variability and experience, as well as to catch any contributory history for each patient.
Several different physicians are a part of the clinic locations or medical groups evaluated in this study. There was no substantial variance between prescribing practices for each clinic when evaluating whether G-CSF medications were used appropriately (Table 2). Each of the clinic locations within this study showed that greater than 80% of patients receiving a G-CSF had it administered appropriately.
Within the 15 patients that were given a G-CSF, there were a total of 36 improper administrations over the study period. Twenty-one of the administrations were pegfilgrastim with the auto-injector, and 15 were pegfilgrastim subcutaneous injections. The amount of inappropriate administrations was associated with an average estimated little over cost of $256,800, utilizing the average wholesale price to estimate the overall cost during the study period.7
We found that colony stimulating factors were used in accordance with the NCCN guidelines in 94.7% of patients evaluated in this study.3 The rates of appropriate use was higher than studies published previously . Hanna et al found that 91.2% of patients were prescribed pegfilgrastim for an appropriate indication.2 Another study by Baig et al.8 found that, among a subset of 400 patients receiving intermediate-risk chemotherapy, appropriate G-CSF use was found in 65% of the patients.8 This study also showed that prophylactic GCSF use was associated with appropriate use in high-risk chemotherapy regimens while they were associated with inappropriate use in intermediate- or low-risk chemotherapy regimens.8
There was no difference in this study between clinic location and inappropriate G-CSF use. In a review conducted by Barnes et al, they found that prescribing practices for including a G-CSF in the treatment regimen varied from 4% to 27% of patients within one management group, suggesting that individual physicians can influence guideline adherence.1 Our study shows that variance is low between locations, with only three locations having appropriate use of G-CSF reported below 90%, according to the criteria used as stated in the methods. Our results suggest that physician practices are similar among multiple locations within the management group in regards to prescribing G-CSFs. Further research could observe the specific patient populations at each location and determine its effect on G-CSF use.
The cost associated with inappropriate use of G-CSF medications was little over $256,800 over the three month period within the NGOC clinics, Wellstar Medical Group, and LaGrange infusion center.7 The formulations associated with inappropriate use were the pegfilgrastim with the auto-injector and the subcutaneous injection. This can be extrapolated to estimate the total yearly cost associated with inappropriate G-CSF use as $1,027,200, utilizing average wholesale pricing to estimate.7 Waters et al.5 estimated the cost of unnecessary G-CSF use as approximately $712,264 over one year, and Hanna et al.2 estimates a potential savings of $600,000 per year if inappropriate use is mitigated.2,5
Pharmacists could play a role in reducing inappropriate G-CSF use in patients who do not meet criteria. A study by Shah, et al, found that clinical pharmacists were involved in 81% of all major drug-specific interventions in the setting of chemotherapy follow up.9 Another study by Nipp et al.10 demonstrated that pharmacist intervention had higher rates of patient vaccinations compared to usual care and that fewer patients had potentially inappropriate medication at week four compared to usual care.10 Fishman et al found that clinical peer-to-peer consultation in oncology may encourage guideline adherence and may lead to a reduction of costs associated with excessive G-CSF use.11 They also found that the risk of febrile neutropenia was not significantly increased when the G-CSF was omitted from therapy in low-risk patients or intermediate-risk patients without risk factors.11 The study conducted by Ignoffo and Knapp12 suggest that pharmacists could be effective in providing services regarding information for pharmacology, pharmacogenomics and oral oncology agents.13 Imamura et al found that the participation of oncology pharmacists allows for safe outpatient chemotherapy while reducing medical costs.13 Other strategies to decrease inappropriate use of G-CSF would be to provide education to the medical staff in the oncology clinics, as peer consultation increases appropriate use, according to the study conducted by Fishman et al.11
There are some limitations of this study. First, the retrospective chart review limited us to only what was recorded in the health system electronic medical record. Second, we were unable to determine if the patient was utilizing a G-CSF for primary or secondary prevention of febrile neutropenia. Finally, we did not evaluate patients that may have been eligible for G-CSF therapy but did not receive it.
We found that over a three-month time period, the majority of G-CSF medication administrations were used in accordance with the NCCN guidelines in the setting of oncology patients within the NGOC, Wellstar Medical Group, and LaGrange infusion center healthcare systems. Future studies could evaluate the inclusion of G-CSF medications in high-risk chemotherapy and determine if patients are being covered appropriately. Another future study could be to determine the efficacy of G-CSFs in preventing episodes of febrile neutropenia within the hospital system.
Tables 1–3
|
Patient demographics (n (%) = 283) |
|
|
Gender |
n (%) |
|
Male |
70 (24.7%) |
|
Female |
213 (75.3%) |
|
Race, n (%) |
|
|
Caucasian |
194 (68.8%) |
|
African American |
61 (21.6%) |
|
Hispanic/Latino |
11 (3.8%) |
|
Asian |
6 (2.2%) |
|
Other or Nondisclosed |
11 (3.8%) |
|
Age, years (SD) |
60.3 (12.8) |
|
Cancer diagnosis n(%) |
|
|
Breast Cancer |
124 (43.8%) |
|
Small Cell Lung Cancer |
25 (8.8%) |
|
Diffuse Large B-cell Lymphoma |
17 (6%) |
|
Pancreatic Cancer |
14 (4.9%) |
|
Uterine Cancer |
14 (4.9%) |
|
Non-Small Cell Lung Cancer |
13 (4.6%) |
|
Colon and Colorectal Cancer |
11 (3.8%) |
|
Prostate Cancer |
9 (3.2%) |
|
Ovarian Cancer |
8 (2.8%) |
|
Hodgkin Lymphoma |
7 (2.5%) |
|
Rectal Cancer |
7 (2.5%) |
|
Non-Hodgkin Lymphoma |
7 (2.5%) |
|
Esophageal and Gastric Cancer |
6 (2.2%) |
|
Peritoneal Cancer |
5 (1.8%) |
|
Bladder Cancer |
2 (0.7%) |
|
Cholangiocarcinoma |
2 (0.7%) |
|
Other* |
12 (4.2%) |
|
Risk of Neutropenia with n (%) chemotherapy |
|
|
High Risk |
148 (52.3%) |
|
Intermediate Risk |
123 (43.5%) |
|
Low Risk |
12 (4.2%) |
|
Medication used n (%) |
|
|
Pegfilgrastim |
279 (98.6%) |
|
Filgrastim |
4 (1.4%) |
Table 1 Baseline characteristics.
|
Site |
Appropriate, n |
Total, n |
Percentage of appropriate use(%) |
|
Douglassville |
11 |
11 |
100% |
|
LaGrange Infusion Center |
4 |
5 |
80% |
|
Roswell |
12 |
14 |
85.7% |
|
Cartersville |
13 |
13 |
100% |
|
Jasper |
10 |
11 |
90.9% |
|
Wellstar Medical Group Gynecology/Oncology |
13 |
15 |
86.7% |
|
Austell |
108 |
109 |
99.1% |
|
Marietta |
58 |
66 |
87.8% |
|
Paulding |
39 |
39 |
100% |
Table 2 Proper use by clinic in the medical group
|
G-CSF agent |
Cost per use (AWP, $) |
Total improper Uses |
Cost times the number of improper doses Improper |
|
Pegfilgrastim with prefilled syringe kit, per injection (Neulasta Onpro) |
7477.27 |
21 |
$157,022.67 |
|
Pegfilgrastim solution, per injection (Neulasta) |
7477.27 |
10 |
$74,772.70 |
|
Pegfilgrastim solution, prefilled syringe, per injection (Udenyca) |
5010 |
5 |
$25,050.00 |
|
Total Cost-Saving Potential during study period |
-- |
36 |
$256,845.37 |
Table 3 Cost of therapy (per injection)7
None.
The authors declared that there are no conflicts of interest.
None.

©2023 Thomas, et al. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and build upon your work non-commercially.
2 7